Patient and public involvement (PPI) in research is the development of an active partnership where researchers, patients, and the public work together to develop research that is relevant and useful to patient and public needs (NIHR Research Design Service, 2018).
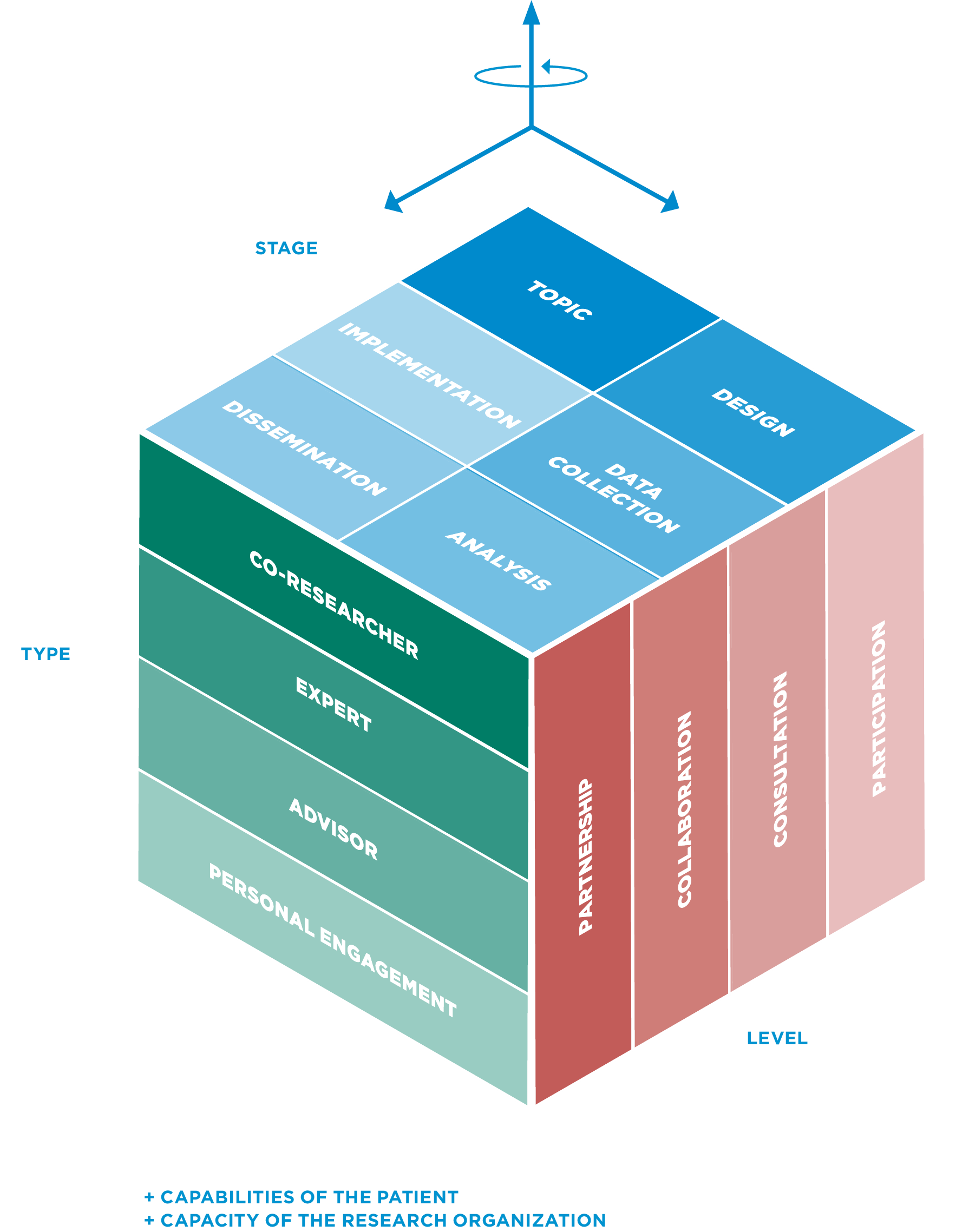
The SCCL-PPI model has been developed based on a literature review that was conducted to identify existing frameworks in cancer research internationally.
Nine models from seven different countries were taken into consideration - leading us to propose a model that is adapted to the specific needs of our local context: French-speaking Switzerland.
To learn more about the development of the SCCL-PPI model, we invite you to take a look at the article we published in the Revue Médicale Suisse.
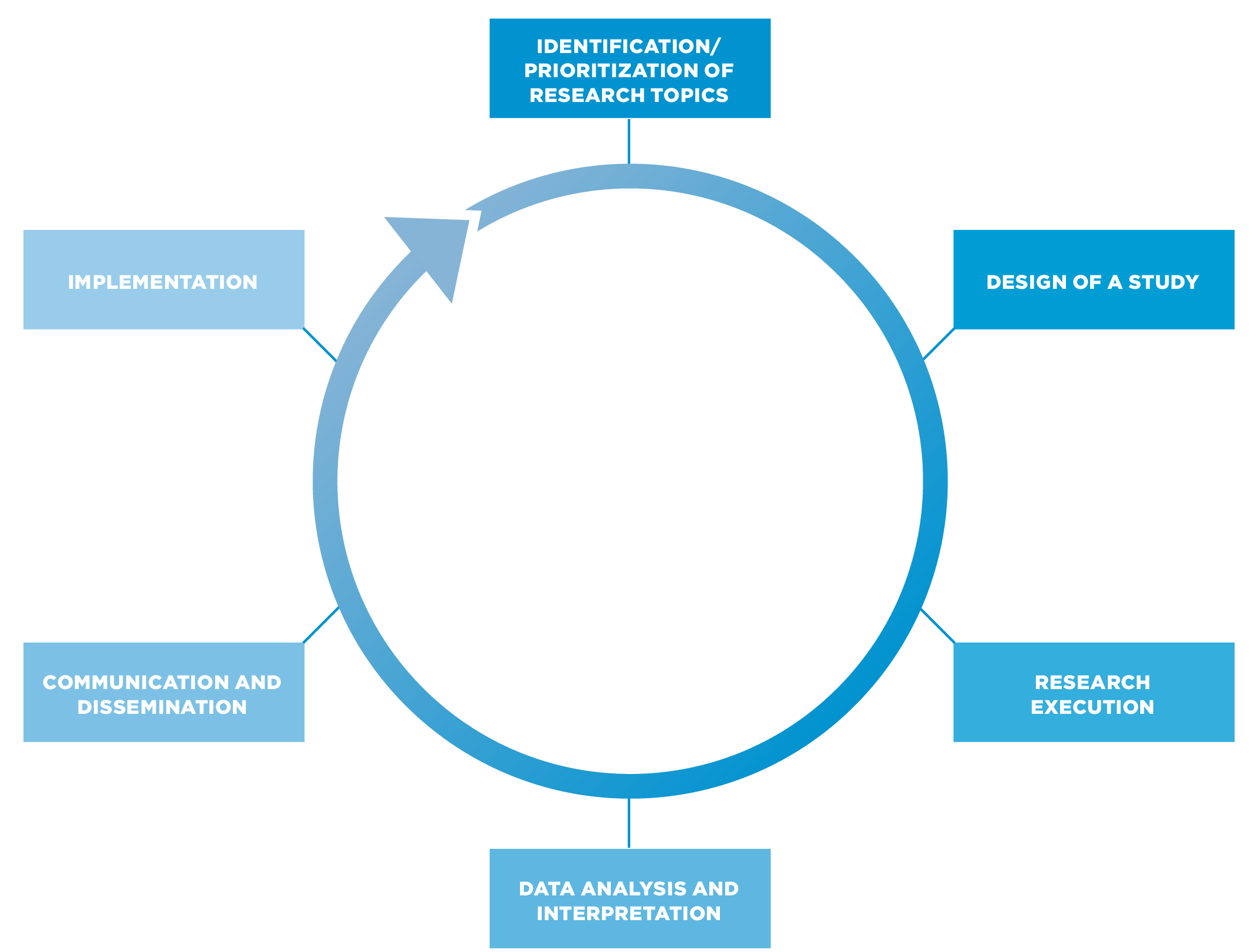
STAGES OF INVOLVEMENT
Patients and the public can be involved at all stages of the research process, and in one or more phases of a research project .
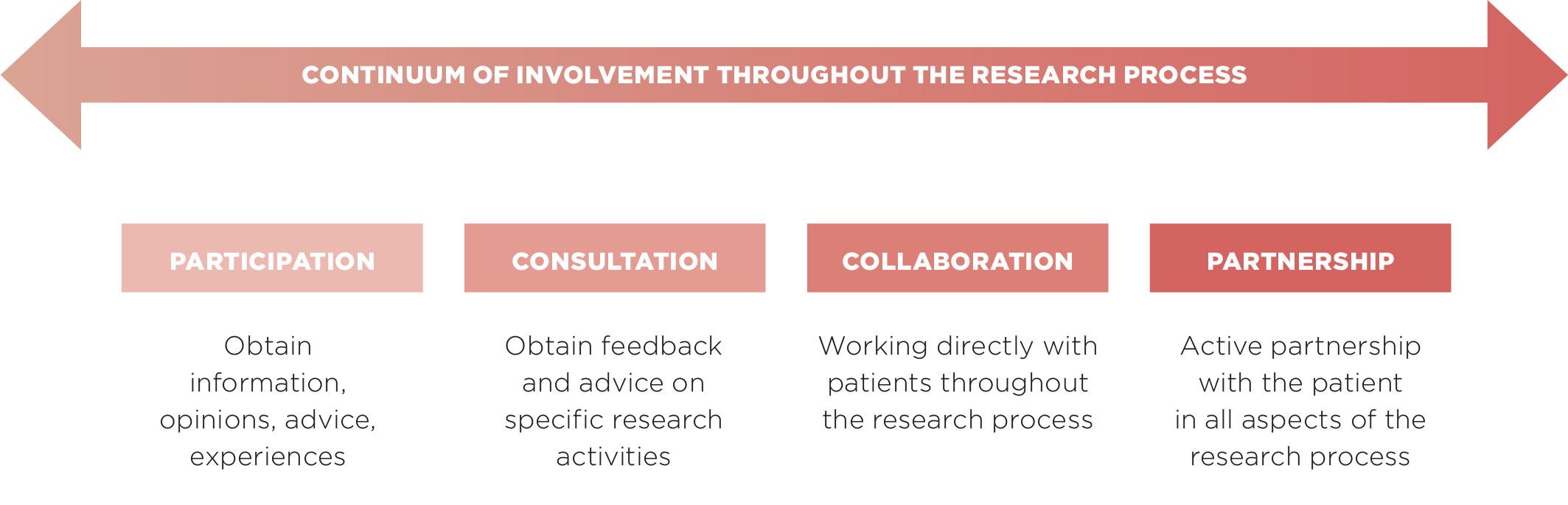
LEVELS OF INVOLVEMENT
In the context of the SCCL-PPI model, and based on the type of research envisioned, we define 4 different levels of patient involvement in research.
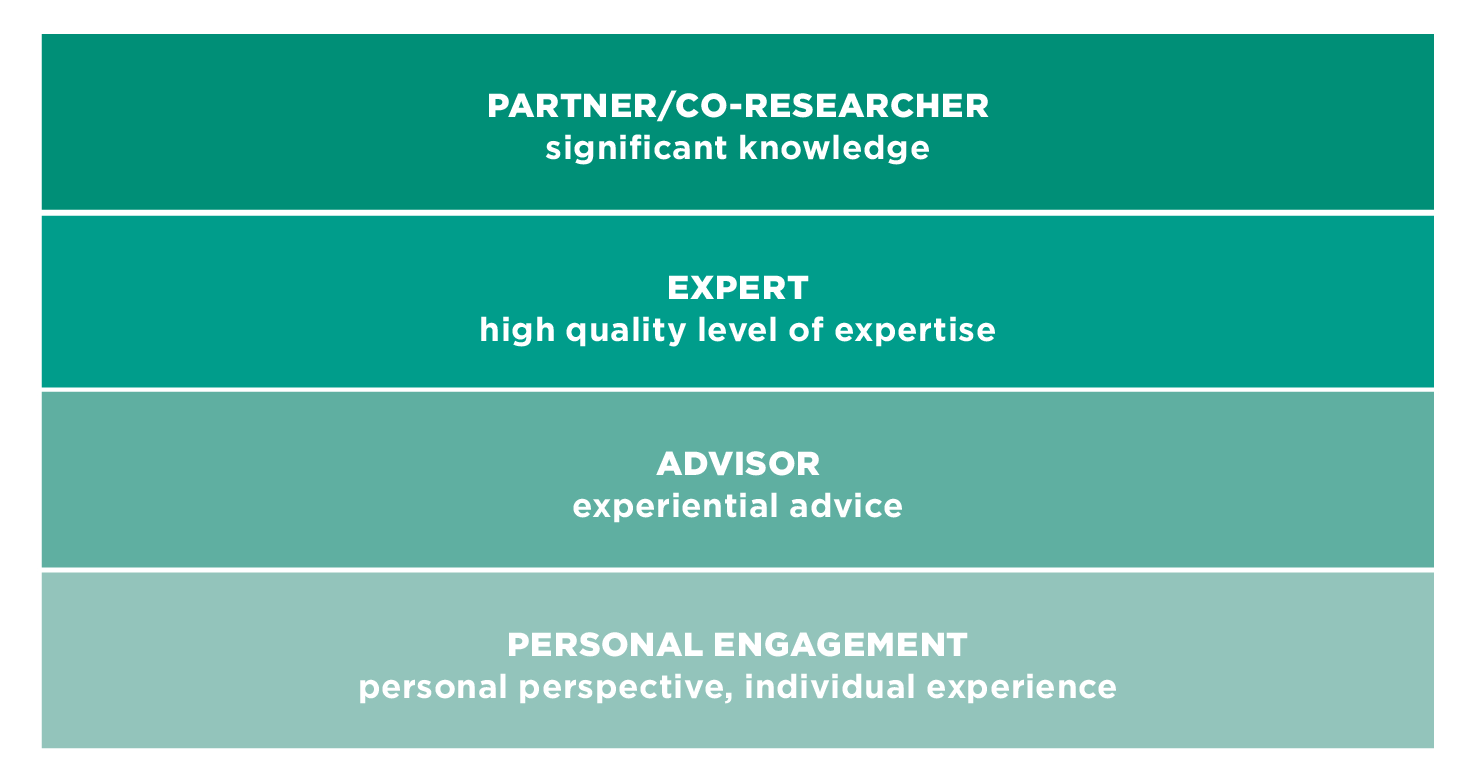
TYPES OF INVOLVEMENT - ROLES
Patients' roles vary according to the contributions a patient is able to offer. To use patient knowledge to the best effect, ways must be found to convert the diversity of individual experience into the closest representation of a typified patient perspective. The patient voice must be a blend of collective and personal experiences.
A way to overcome this is to include more than one patient and covering more than one type of involvement.
CAPABILITIES & CAPACITIES
Furthermore, the decision regarding the stage, level, and type of involvement requires a careful evaluation of the capabilities of the patients as well as the capacity of the research group or organization.
